Why the UK Pharma Industry Could be a Casualty in the Conflict Over Greenland
A deal in December promised more money for innovative medicines, potentially encouraging increased investment in UK pharma facilities. The tensions in the Arctic mean that’s now up in the air.
Trump knows how to keep people guessing. The shock new tariffs announced against European countries that the President believes are thwarting his desire to buy the territory continue the US “unwinding its own global order”.
Under Trump, the norms that governed that order are unravelling, including those related to trade. Following last year’s “Liberation Day” tariffs, UK producers were adjusting to the new normal. Some may even have seen a comparative advantage in the lower 10% tariff rate UK manufacturers faced, compared with some of their international competitors.
The US trade deal announced in May was fairly sketchy and did little to improve things for most, even if it brought some relief for the likes of the car manufacturers. But it, again, began to give some clarity over future trading conditions. It also included a commitment to preferential treatment for UK pharmaceuticals, should the US impose tariffs in the future, which has since been built upon.
Now this is all potentially up in the air, with Trump threatening a (presumably additional) 10% tariff on goods from the UK, as well as Denmark, Norway, Sweden, France, Germany, the Netherlands and Finland from 1 February. Given the difficulties of targeting individual European nations in the single market, it would probably mean tariffs for the whole of the EU.
From 1 June, the President threatens that these will rise to 25% if there is no agreement. “This tariff will be due and payable until such time as a deal is reached for the complete and total purchase of Greenland,” the President wrote.
Supply Chain Resilience for Vital Drugs
The method in Trump’s madness is not always clear, but when it comes to pharma, it is possible to discern his aims. They have largely been twofold.
The first, as we’ve touched on before, is the administration’s concern – shared widely across the political spectrum – over pharma supply chain fragility. The case for the defence is that Trump is trying to ensure more resilient supplies of key drugs by reducing dependence on production in China and India.
“Essentially, Washington is signalling that the era of America’s unlimited drug imports is ending. And that’s good news because the United States long ago surrendered control of its pharmaceutical supplies to overseas factories with weak oversight and chronic safety failures,” writes one supportive columnist. So while the President has been hiking tariffs elsewhere, he was, at least in December, seeking to cut tariffs for UK pharmaceuticals.
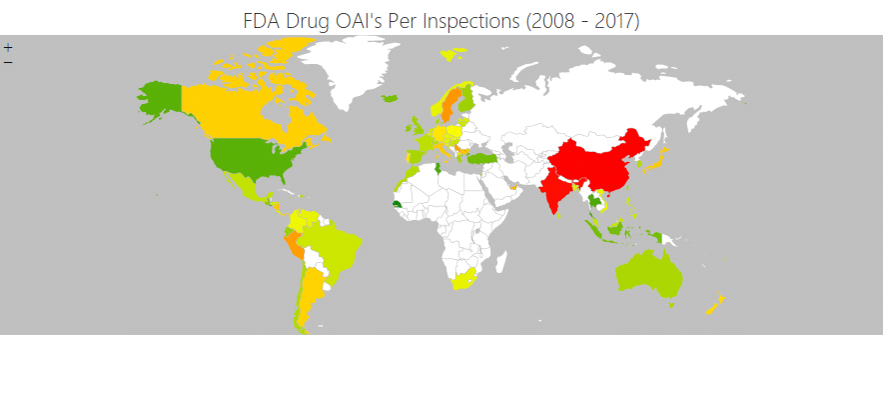
There’s some truth in this. The US – and world – is heavily dependent on China (and India to a lesser extent) for active pharmaceutical ingredients. It’s also true that standards overseas have not been as high. That’s been the case for years. A study led by Intersys a decade ago found that more than two-thirds of life sciences shortages were linked to notices issued by the US FDA, often on manufacturers in China and India.
However, it’s also true, as we’ve again noted, that this isn’t easy to fix. The low margins on generics mean that onshoring looks unlikely in many cases. As some noted when the Liberation Day tariffs were announced last year, the costs of relocation for drug companies are far greater than the impact of most tariffs envisaged.
More Money for Innovative Medicines
From Trump’s point of view, trade policy and tariffs around pharma are not just about securing supply chains, however. He’s also framed them around fairness for American manufacturers.
That’s why the agreement in principle with the UK at the start of December on pharma, following last May’s trade deal, was specifically on pharmaceutical pricing. This was the cost attached to cutting tariffs for UK pharmaceuticals.
“President Trump is the first American President to work with US trading partners to ensure fair payment internationally for innovative pharmaceuticals and pharmaceutical ingredients. For too long, American patients have been forced to subsidise prescription drugs and biologics in other developed countries by paying a significant premium for the same products in ours,” said Ambassador Jamieson Greer, the US Trade Representative.
“Today, the United States and the United Kingdom announce this negotiated outcome pricing for innovative pharmaceuticals, which will help drive investment and innovation in both countries. The Trump Administration is reviewing the pharmaceutical pricing practices of many other US trading partners and hopes that they will follow suit with constructive negotiations.”
The agreement means that the UK would “reverse the decade-long trend of declining National Health Service expenditures on innovative, life-saving medicines”, and increase the net price it pays for new medicines by 25%. In practice, the rebate that drug firms pay to the NHS, a proportion of revenues from new branded medicine sales, was cut from 22.9% to 14.5%. (The deal actually saw the British government agree to cap the rebate rate for newer drugs at a maximum of 15% until 2028.)
Saving Costs of Cutting Pharma Investment?
In some ways at least, this deal was a win-win for the UK, and certainly for UK pharma. The exemption from US tariffs was an obvious goal for the pharma industry. But the cost extracted for the concession would probably also benefit the UK long term, as Fierce Pharma explains.
“Before the US stepped in, the UK government and the biopharma industry had deadlocked on a new VPAG [rebate] rate. While the government was trying to contain overall healthcare costs, the industry argued that the proportion of those expenditures allocated to novel medicines was shrinking and lagging behind other developed countries.”
It notes the warning from the Association of the British Pharmaceutical Industry in June that high rebate rates meant the UK could lose £11 billion in pharma R&D by 2033, and see fewer new medicine launches in the NHS.
That’s not an idle threat. We’ve already seen several pharmaceutical companies, including British companies like AstraZeneca and GSK, scale down investment in UK facilities, with the UK’s unwillingness to pay for innovative medicines a constant complaint. Last August, Norvartis’s UK head, Johan Kahlstrom, said the country was “largely uninvestable”.
The December deal would not have changed that overnight. In an interview a little more than a week after the deal, GSK’s chief executive Dame Emma Walmsley reiterated that the US remained the key market for pharma.
“No one should be deluded that the UK is going to be a massive scale market, a domestic market, but it can be an exporter of innovation in life sciences,” she told the BBC. “For GSK, it's 2% of our sales are here. More than 50% are in the US.”
Nevertheless, in edge cases, a greater willingness to pay for innovative medicines may have secured more investment in UK facilities.
For once in negotiations with Trump, this seemed like a win-win. If it falls foul of the row over Greenland, it will be everyone’s loss.