Industrial Strategies, Supply Chains, Life Sciences and Advanced Manufacturing
The government’s industrial strategy has been delayed and may be derailed by competing priorities and a lack of funding. However, there are lessons businesses across different industries can take from it already.
After seven years, what’s a few more months? The government’s industrial strategy, announced in October 2024, but a few months after Labour won the July election, was broadly welcomed. The first in seven years, the plan was to be central to the “number one mission” of the new government, according to Rachel Reeves: Growth.
“This industrial strategy will provide a launchpad for businesses. It provides the firm foundation for investment that businesses have told us they need,” she wrote. “This government believes it is our role to provide the certainty that inspires confidence, allowing businesses to plan not just for the next year, but for the next 10 years and beyond.”
The Institute of Government was welcoming but cautious. It said that the strategy avoided obvious pitfalls but warned, “That which is quickly broken is not speedily fixed.”
Now, it seems the fix must wait a while longer. Just like the growth the government wants to see, the industrial strategy has yet to materialise. In mid-February, it was reported that the strategy’s publication had been pushed back to June, around the time of the government spending review.
The same month also saw the conclusion of the House of Commons’ Business and Trade Committee’s inquiry into the Industrial Strategy proposals. It will now make recommendations for the Comprehensive Spending Review to be complete late Spring.
The delay means it will be some time before businesses can see precisely what proposals from the Green Paper put out for consultation make it into the White paper outlining the concrete policies.
However, aware no doubt that business confidence is at a low ebb, reports of the delay to the strategy also reveal that publication of strategies for particular sectors may be brought forward. Among them is life sciences.
Services, manufacturing and supply chains
Life sciences was among eight “growth-driving sectors” identified in the Green Paper as a focus of the industrial strategy.
“They include the services and manufacturing industries that present the greatest opportunity for output and productivity growth over the long-term,” Reeves’ foreword to the report stated. The complete list includes a range of other advanced, multi-stage production industries, including life sciences:
- Advanced manufacturing
- Clean energy industries
- The creative industries
- Defence
- Digital and technologies
- Financial services
- Professional and business services
- Life sciences
The “next stage of development of the industrial strategy” (whether this means the White Paper or further down the line is unclear) will see the government further prioritise various subsectors of each sector “where there is evidence that policy can address barriers to growth.”
The consultation paper also identified some of the barriers the government will be looking at. These include skills and recruitment, research and development, data, infrastructure, planning, competition, regulation, grid connections and energy prices.
Moreover, the paper also states that the industrial strategy will seek to “reduce supply chain and other vulnerabilities in growth-driving sectors” that could harm their long-term prospects. The government is also establishing a supply chains taskforce to identify supply chains critical to the UK’s economic security and resilience and examine their vulnerabilities.
Boost for Oxford-Cambridge pharma
Other announcements have underscored the government’s good intentions for growth. In January, for example, the government hired a new head for the Competition and Markets Authority: former Amazon UK head Doug Gurr, an appointment seen as emphasising the government’s commitment to growth and by unions as a “slap in the face to workers”.
Also in January, Reeves announced backing for a third runway at Heathrow – a desire of many pro-growth campaigners for two decades. Significantly for life sciences, in the same speech, Reeves announced backing for the expansion of the Oxford-Cambridge corridor, an area she said had the potential to be “Europe’s Silicon Valley” but also houses a cluster of UK life science businesses. Plans would include new transport links and new developments and other infrastructure.
Nevertheless, even the most optimistic will concede that identifying barriers to growth will be easier than breaking them down.
Some will see contradictions within the government’s other policy priorities. While investigating how UK energy costs – among the highest in the world – are hitting growth, the Green Paper also promises to “support net zero”, which many blame for contributing to this.
The reaction from environmental activists to Reeves’ backing for a third Heathrow runway highlighted some of the tensions, too. Heathrow is anyway a powerful lesson in good intentions on growth not being sufficient: Labour’s current support for a third runway echoes the policy of a previous Labour government’s announcement backing the project – when Tony Blair was leader.
Bad omens: AstraZeneca’s Speke plant
It’s a similar story when it comes to employment.
“Jobs will be at the heart of our modern industrial strategy,” promised Reeves in the Green Paper. However, the increase in Employers’ National Insurance contributions coming into effect in April is among the factors said to be leading businesses to brace for the largest wave of redundancies in a decade outside of the pandemic, according to the CIPD.
The biggest block to tackling the barriers to growth, though, may just be the absence of growth itself. With the government finances still strained from the costs of Covid, there’s a limit to the financial help it can put forward.
We’ve arguably already seen the impact in January withAstraZeneca scrapping its plans for a £450 million vaccine plant it was to build in Liverpool suburb Speke. Agreed under the last government, the deal fell through in part, according to the company, because of less favourable funding offered by Kier Starmer’s team.
“Several factors have influenced this decision, including the timing and reduction of the final offer compared to the previous government's proposal,” a spokesperson for the pharma giant told Sky News.
Partnership in practice: Supply chain strategies
There is, of course, still much the government can do beyond offering financial incentives for inward investment. Many of its proposals would see the government simply getting out of the way, loosening regulations and removing constraints. Time will tell if it’s committed to do so.
In any case, though, it’s clear that the greatest help will be for those prepared to help themselves.
The Green Paper insists the strategy is based on a partnership: “This approach is one that seeks to place private business, entrepreneurship, and innovation at its heart, supported by governments playing a strategic and coordinating role beyond the fundamentals of upholding the rule of law and macroeconomic stability. It is an approach of partnership between government, businesses, and workers, working together to create the conditions for sustained and long-term growth across the economy.”
We see this reflected in the opportunities identified for strengthening supply chains. The paper offers several ideas. One is “promoting productivity and growth in small businesses across our economy, particularly in supply chains across our growth-driving sectors.” If achieved, that should offer all businesses in the targeted sectors opportunities to strengthen resilience.
Another, primarily up to the government, is strengthening international partners to reduce critical dependencies, especially for critical goods. It names critical minerals, semiconductors, and batteries.
Finally, it also suggests “improving data maturity in businesses to help businesses to do more with data… and supporting businesses in the way data is used across supply chains.”
Technology beyond pharma
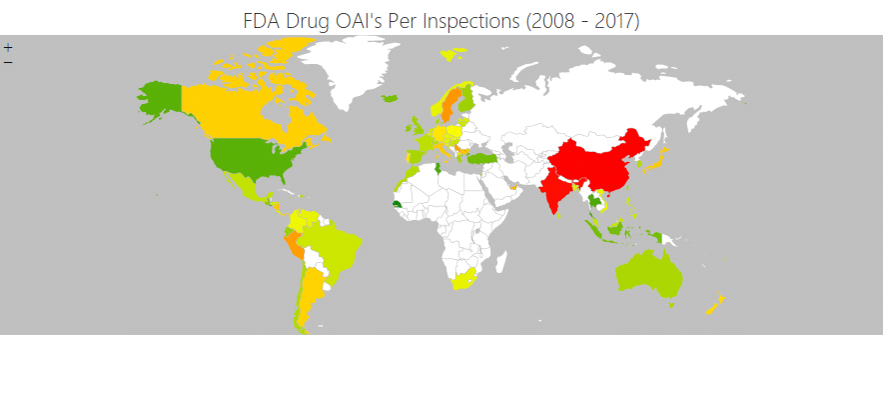
No doubt there has been a significant government focus on life sciences when it comes to the issue of supply chains. One example of success the Green Paper notes, for instance, is the delivery of 13,437 ventilators to the NHS during Covid by implementing an end-to-end supply chain in 1.5 weeks.
But, as the paper correctly implies, many supply chain issues facing pharma are common across industries. Consequently, the opportunities to transform them through technology are also widespread.
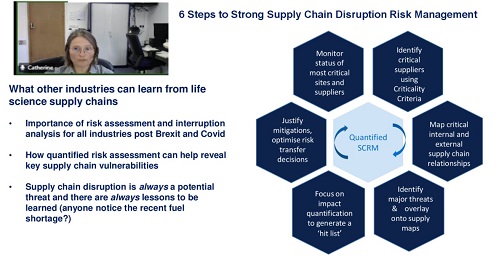
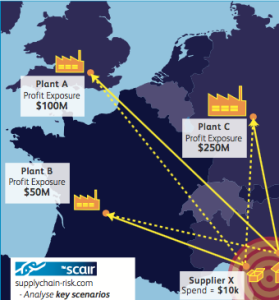
The lessons we’ve long preached on promoting visibility of the connections and value at risk across the supply chain that are applicable to life sciences equally apply to other sophisticated, multi-stage production industries with complex, geographically dispersed supply chains.
That includes other sectors targeted by the industrial strategy, such as advanced manufacturing and parts of technology. Whatever comes out in the final proposals for the industrial strategy – and when – this lesson is one that businesses can take and start acting on now.